Strawberry does not taste like strawberry without a touch of cooked sugar.
It sounds contradictory. But it is basic flavor chemistry.
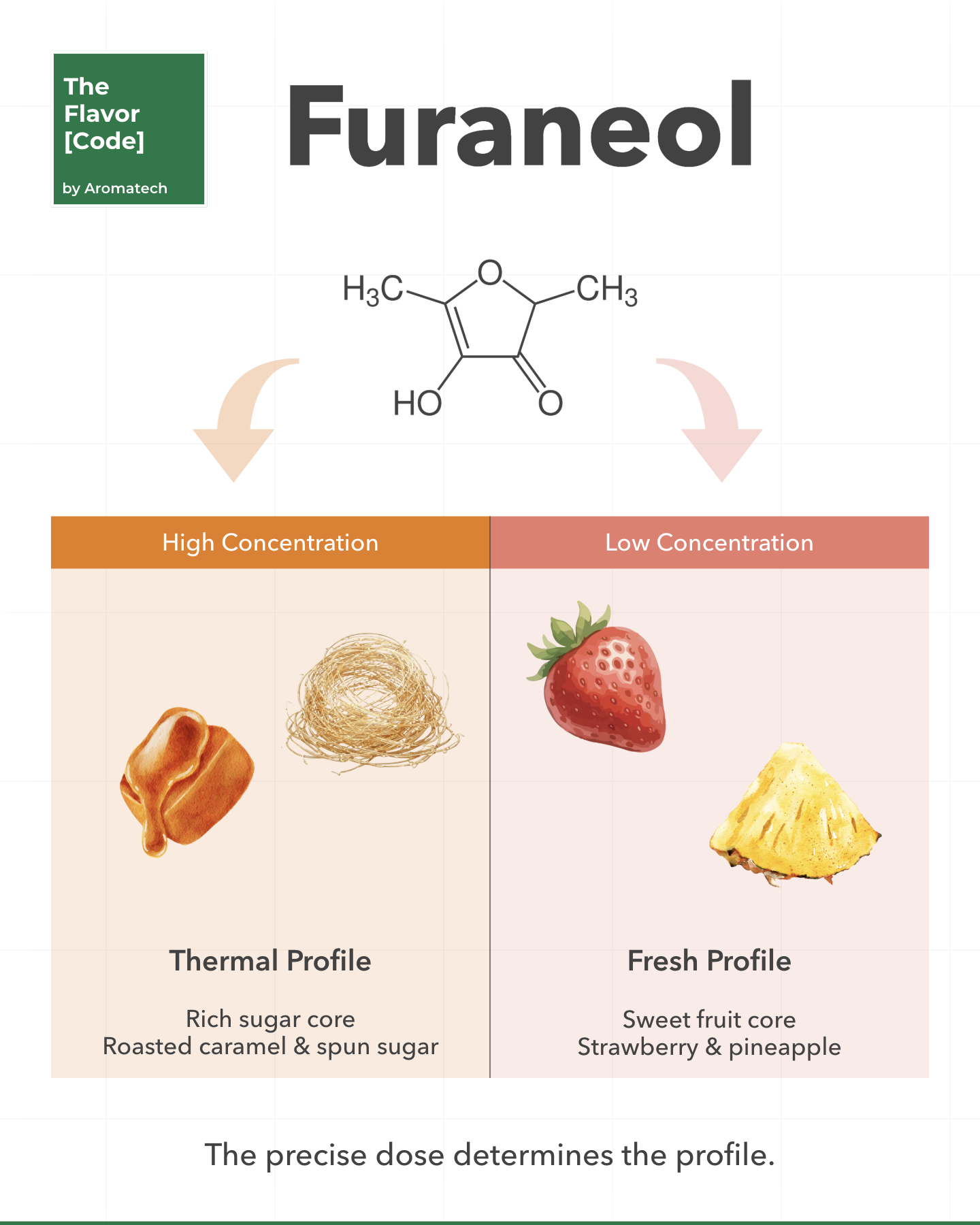
At the center of an authentic fruit profile is a molecule with a split personality. Furaneol.
In the lab, we look at it as a shapeshifter.
At high concentrations, spun sugar and roasted caramel.
At low concentrations, the unmistakable sweet core of fresh strawberries and pineapples.
Biologically, a strawberry builds it through complex enzymatic pathways as it ripens.
Thermally, it forms when reducing sugars like rhamnose react with amino acids under heat.
This duality makes it a “Sweet Bridge” in product formulation.
But here is the pain point we often see in beverage development.
You want a fresh, clean strawberry sparkling water.
So you load up on C6 volatiles for that green, leafy bite.
Then you add Furaneol to build the ripe, jammy body.
And suddenly, your fresh water tastes like a baked pastry.
The beverage matrix is amplifying the thermal side of the molecule.
Cutting the Furaneol will not fix it. You have to manage the bridge.
🔬 Control the threshold. Furaneol has an incredibly low odor detection limit. A fraction of a part per million changes the entire system.
🍓 Pair it with its methyl ether derivative (DMMF). This creates a more authentic, less heavy fruit anchor.
🍋 Adjust acidity. A sharp drop in pH cuts the heavy caramel notes and pulls the profile back toward fresh fruit.
Knowing the molecule is easy.
But knowing how it behaves under pressure is what makes the flavor work.