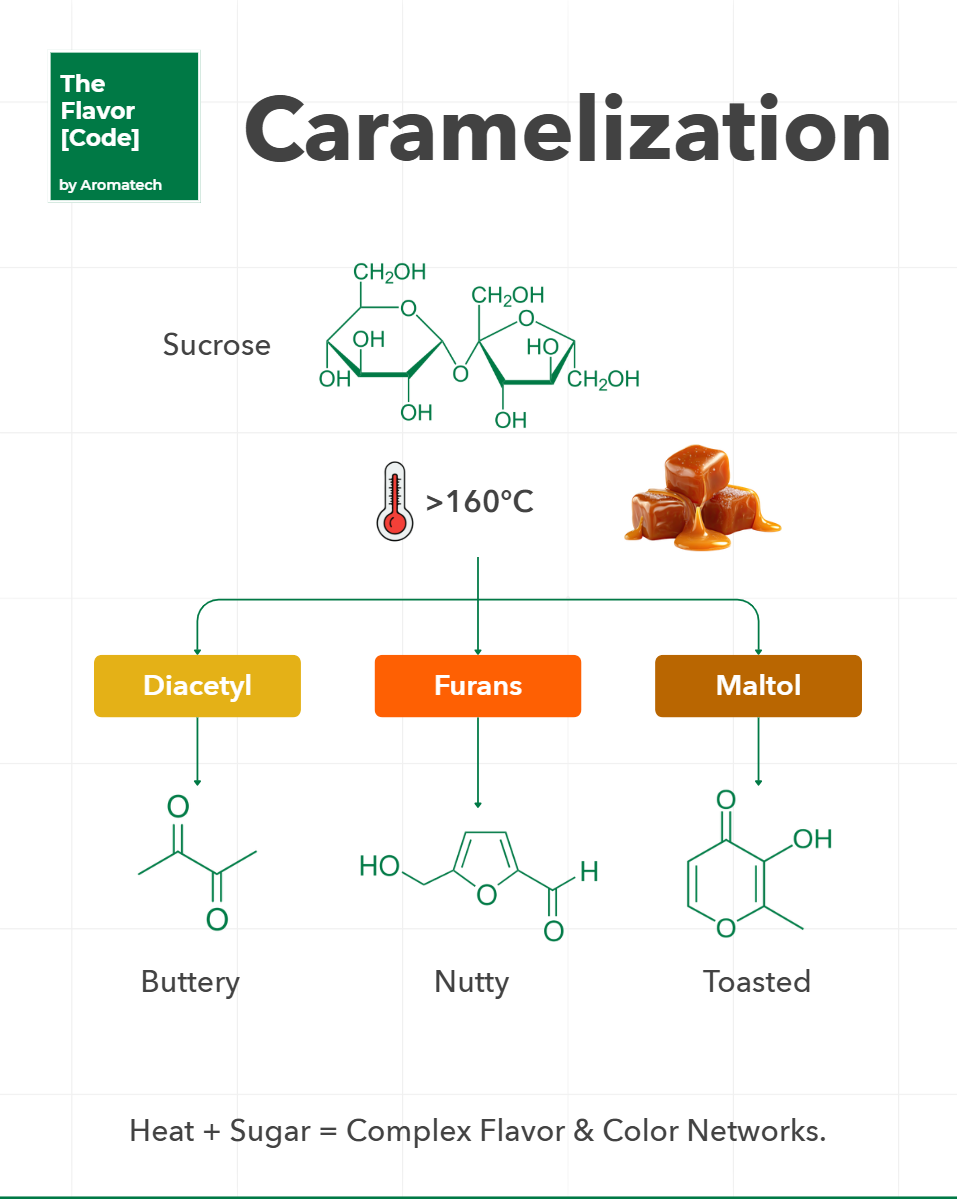
Sugar does not just melt.
It undergoes apparent melting, a kinetic thermal decomposition.
It shatters.
When you push sucrose past 160C the ring structures break down.
This is the raw chemistry of caramelization.
It is a chaotic cascade of dehydration and fragmentation.
And it creates the foundation for some of the most complex profiles in flavor chemistry.
Welcome to The Flavor Code.
Today we are looking at the molecular breakdown of burning sugar.
The process forces carbohydrates to reassemble into hundreds of volatile compounds. But controlling this reaction on the bench is notoriously difficult.
Push the heat too long and you get intense bitterness.
Drop the pH and the entire sensory profile shifts.
Here are the three primary volatile structures you generate.
🧪 Furans deliver the signature toasted and nutty aromatics.
They form when sugar undergoes severe dehydration and ring closure.
🧬 Maltol provides the sweet and baked sugar perception.
It acts as a powerful flavor enhancer even at low parts per million.
🔬 Diacetyl emerges from the fragmentation of the sugar skeleton.
It gives the system that heavy and creamy butter perception.
So you have to balance these three competing pathways.
A slight temperature fluctuation ruins the batch.